
One typical math equation you will certainly see is the estimation of official cost. Nevertheless, this training course is based on understanding concepts and the technique of response device series. Again, I think this goes back to applying the previous years’ research methods to the brand-new and hard subject material. This concern frightens many students more significant than the initial inquiry. How Can You Have An Entire Training Course With Just Five Mathematics Equations? The ordinary organic chemistry trainee is in Charge of up to five mathematical formulas throughout the whole course. My response and article emphasis instead gets on the academic or talk portion of the concern. Gram to mole and associated estimations do not alter. After all, many of the measurements and computations are used from the previous year of introductory chemistry. This suggestion leaves out the math that you must recognize and utilize when working on your natural laboratory reports. The surprising answer I have for them, and for you, is that there is very little math in this course. “Just How Much Math Is There In Organic Chemistry?” They ask this because they are looking to continue their important chemistry research study and technique behaviours. Formula For Formal ChargeĪs a natural chemistry tutor, I commonly get this question from my pupils that have just finished a math-filled year of general chemistry. Instead, you take a look at an atom, count the number of electrons straight affixed, as well as deduct this number from the ground-state valence electrons typically discovered on a neutral bit of this type. I’m stating the same point as the complicated formula above because this includes the single electrons located in pairs, As Well As half of the bonding electrons.īut counting this way, you don’t find yourself doing numerous estimations. HAS = the number of electrons directly attached to the atom in your lewis framework. NEED TO = variety of valence electrons the atom must have, located on the periodic table That’s all you have to remember, now let’s clarify. 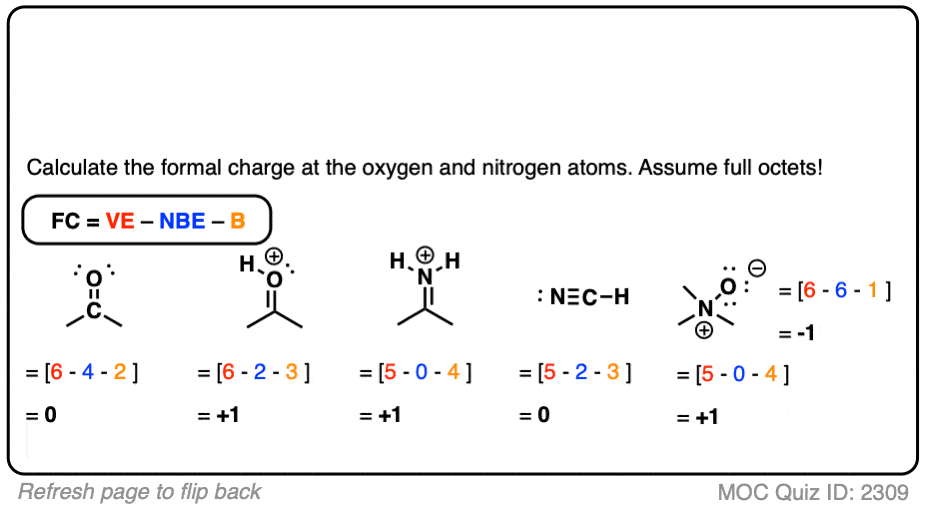 
We then include half the bonding (or directly connected electrons) to the single sets. Every bond has 2 electrons, which indicates that if we divide the array of bonds by 2, we get the bonding electron associated with that atom. this component we keep as is, we require to count affixed electrons The next component is the complicated one. You can find this number by counting the atoms team on the periodic table. This refers to the number of valence electrons located in the ground state atom. Yet if you understand the formula, you can simplify it to resolve in secs. If you’re overcoming a response and a need to recognize a fee QUICKLY, you do not have time to resolve the above. Nevertheless, this training course is based on understanding concepts and the technique of response device series.The next component is the complicated one.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
